Pharma ADMET Testing Market to Reach $18,062.89 Bn by 2026 as Advanced ADMET Technologies Transform Drug Development
The Business Research Company's Pharma ADMET Testing Market Report 2026 – Market Size, Trends, And Global Forecast 2026-2035
LONDON, GREATER LONDON, UNITED KINGDOM, March 9, 2026 /EINPresswire.com/ -- "Pharma ADMET Testing Market to Surpass $18 billion in 2030. Within the broader Healthcare Services industry, which is expected to be $10,759 billion by 2030, the Pharma ADMET Testing market is estimated to account for nearly 0.2% of the total market value.
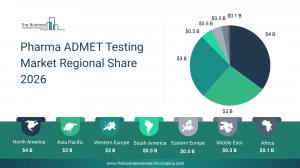
Which Will Be the Biggest Region in the Pharma ADMET Testing Market in 2030
North America will be the largest region in the pharma ADMET testing market in 2030, valued at $5,863 million. The market is expected to grow from $3,668 million in 2025 at a compound annual growth rate (CAGR) of 10%. The rapid growth can be attributed to the rising public scrutiny of drug safety and shift toward complex biologics and novel modalities.
Which Will Be The Largest Country In The Global Pharma ADMET Testing Market In 2030?
The USA will be the largest country in the pharma ADMET testing market in 2030, valued $5,283 million. The market is expected to grow from $3,336 million in 2025 at a compound annual growth rate (CAGR) of 10%. The rapid growth can be attributed to the growth in multi-omics integration and rising public scrutiny of drug safety.
Request a free sample of the Pharma ADMET Testing Market report:
https://www.thebusinessresearchcompany.com/sample_request?id=12024&type=smp&utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Mar_PR
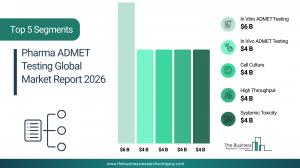
What will be Largest Segment in the Pharma ADMET Testing Market in 2030?
The pharma ADMET testing market is segmented by testing type into in vivo ADMET testing, in vitro ADMET testing and in silico ADMET testing. The in vitro ADMET testing market will be the largest segment of the pharma ADMET testing market segmented by testing type accounting for 52% or $9,436 million of the total in 2030. The in vitro ADMET testing market is supported by demand for cost-efficient early-stage toxicity screening, increasing adoption of human-cell–based assays for better predictive accuracy, rising shift toward reducing animal testing, growing use of high-throughput laboratory techniques, expanding need for rapid compound filtration during lead optimization, and the ability to detect organ-specific toxicities with controlled experimental settings.
The pharma ADMET testing market is segmented by technology into cell culture, high throughput, molecular imaging and OMICS technology. The high throughput market will be the largest segment of the pharma ADMET testing market segmented by technology, accounting for 37% or $6,655 million of the total in 2030. The high throughput market will be supported by growing need to screen large compound libraries efficiently, rising adoption of automated platforms to shorten drug development timelines, increasing focus on parallel testing for multiple ADMET parameters, expanding pharmaceutical R&D investments, rising dependency on miniaturized assays for cost reduction, and the ability to accelerate lead optimization with higher accuracy.
The pharma ADMET testing market is segmented by application into systemic toxicity, renal toxicity, hepatotoxicity, neurotoxicity and other applications. The systemic toxicity market will be the largest segment of the pharma ADMET testing market segmented by application, accounting for 35% or $6,328 million of the total in 2030. The systemic toxicity market will be supported by rising requirements to evaluate whole-body safety before clinical trials, increasing emphasis on identifying adverse multi-organ effects, growing use of integrated testing systems for broad toxicity screening, expanding regulatory scrutiny around systemic exposure limits, rising demand to understand dose-dependent reactions, and the need to identify early-stage safety risks across organ systems.
What is the expected CAGR for the Pharma ADMET Testing Market leading up to 2030?
The expected CAGR for the pharma ADMET testing market testing leading up to 2030 is 11%.
What Will Be The Growth Driving Factors In The Global Pharma ADMET Testing Market In The Forecast Period?
The rapid growth of the global pharma ADMET testing market leading up to 2030 will be driven by the following key factors that are expected to reshape pharmaceutical ADMET quality assurance, drug development, and industrial processes worldwide.
Expansion Of High-Throughput Screening (HTS) Platforms- The expansion of high-throughput screening (HTS) platforms will become a key driver of growth in the pharma ADMET market by 2030. As drug developers increasingly adopt HTS technologies to rapidly evaluate large libraries of compounds for absorption, distribution, metabolism, excretion and toxicity (ADMET) properties, the demand for scalable and efficient ADMET testing services will rise. These platforms enable simultaneous early-stage screening of hundreds to thousands of compounds, reducing time-to-data, lowering costs per candidate, and enabling faster prioritization of safe and efficacious drug leads. As pharmaceutical and biotech companies accelerate discovery cycles and broaden their pipelines, especially with growing interest in small molecules and biologics, the need for robust, automated ADMET testing supported by HTS infrastructure will expand rapidly, thereby driving growth of the pharma ADMET testing market. As a result, the expansion of high-throughput screening (HTS) platforms is anticipated to contributing to a 1.5% annual growth in the market.
Growth In Multi-Omics Integration - The growth in multi-omics integration will emerge as a major factor driving the expansion of the market by 2030. As drug developers increasingly combine insights from genomics, proteomics, metabolomics and transcriptomics, there is a growing need for advanced ADMET testing frameworks capable of interpreting complex biological interactions. Multi-omics integration supports deeper understanding of metabolic pathways, toxicity mechanisms and individual variability in drug response, enabling more accurate prediction of safety and efficacy profiles. As pharmaceutical companies adopt these multi-layered analytical approaches to enhance decision-making, reduce late-stage failures and strengthen precision-medicine initiatives, the demand for comprehensive, data-rich ADMET testing solutions will rise substantially, thereby driving the growth of the pharma ADMET testing market. As a result, the growth in multi-omics integration is anticipated to contributing to a 1.0% annual growth in the market
Rising Public Scrutiny Of Drug Safety - The rising public scrutiny of drug safety within digital manufacturing processes will serve as a key growth catalyst for the market by 2030. Growing awareness among patients, regulators and advocacy groups about adverse drug reactions and long-term safety has increased pressure on pharmaceutical companies to ensure rigorous safety evaluation before drug approval and market release. This heightened scrutiny drives demand for comprehensive ADME, toxicology and pharmacokinetic testing to detect potential adverse effects early and to ensure regulatory compliance. As transparent reporting systems and real-world safety monitoring become more widespread, the requirement for robust preclinical and translational ADMET testing will grow, thereby fueling expansion in the pharma ADMET testing market. Therefore, this rising public scrutiny of drug safety across digital manufacturing operations is projected to supporting to a 0.8% annual growth in the market.
Shift Toward Complex Biologics And Novel Modalities - The shift toward complex biologics and novel modalities will become a significant driver contributing to the growth of the market by 2030. As pharmaceutical research increasingly embraces advanced biologic drugs, gene therapies, antibody–drug conjugates, and other novel modalities, there is rising demand for specialized ADMET and safety assessment workflows tailored to the unique pharmacokinetic and metabolic behaviors of these therapies. These complex biologics often require customized absorption, distribution, metabolism and excretion studies, as well as in-depth toxicity profiling, to ensure safe and effective clinical translation. As more drug developers focus on these next-generation therapeutic formats, the need for robust, modality-specific ADMET testing services will expand substantially, thereby driving growth in the pharma ADMET testing market. Consequently, the shift toward complex biologics and novel modalities is projected to contributing to a 0.5% annual growth in the market.
Access the detailed Pharma ADMET Testing Market report here:
https://www.thebusinessresearchcompany.com/report/pharma-admet-testing-global-market-report?utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Mar_PR
What Are The Key Growth Opportunities In The Pharma ADMET Testing Market in 2030?
The most significant growth opportunities are anticipated in the in vitro ADMET testing market, the high-throughput ADMET screening market, and the pharmaceutical systemic toxicity and ADMET evaluation market. Collectively, these segments are projected to contribute over $10 billion in market value by 2030, driven by increasing drug development pipelines, growing emphasis on early-stage toxicity screening, and regulatory pressure to reduce late-stage clinical failures. This surge reflects the accelerating adoption of predictive laboratory technologies that enable faster compound evaluation, improved safety profiling, and cost-efficient pharmaceutical R&D, fueling transformative growth within the broader ADMET testing industry.
The in vitro ADMET testing market is projected to grow by $4,333 million, the high-throughput ADMET screening market by $3,141 million, and pharmaceutical systemic toxicity and ADMET evaluation market by $2,627 million over the next five years from 2025 to 2030.
The Business Research Company (www.thebusinessresearchcompany.com) is a leading market intelligence firm renowned for its expertise in company, market, and consumer research. We have published over 17,500 reports across 27 industries and 60+ geographies. Our research is powered by 1,500,000 datasets, extensive secondary research, and exclusive insights from interviews with industry leaders.
We provide continuous and custom research services, offering a range of specialized packages tailored to your needs, including Market Entry Research Package, Competitor Tracking Package, Supplier & Distributor Package and much more
Disclaimer: Please note that the findings, conclusions and recommendations that TBRC Business Research Pvt Ltd delivers are based on information gathered in good faith from both primary and secondary sources, whose accuracy we are not always in a position to guarantee. As such TBRC Business Research Pvt Ltd can accept no liability whatever for actions taken based on any information that may subsequently prove to be incorrect. Analysis and findings included in TBRC reports and presentations are our estimates, opinions and are not intended as statements of fact or investment guidance.
The Business Research Company
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email: info@tbrc.info"
Oliver Guirdham
The Business Research Company
+44 7882 955267
info@tbrc.info
Visit us on social media:
LinkedIn
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.